Heat Pumps and Refrigerators
Heat Pumps and RefrigeratorsIn a heat engine, the direction of energy transfer is from the hot reservoir to the cold reservoir, which is the natural direction. The role of the heatthe engine i...
INTRODUCTION
Simply heat is the sum of kinetic energy of atoms or molecules. In thermodynamics, heat means energy which is moved between two things when one of them is hotter than the other.
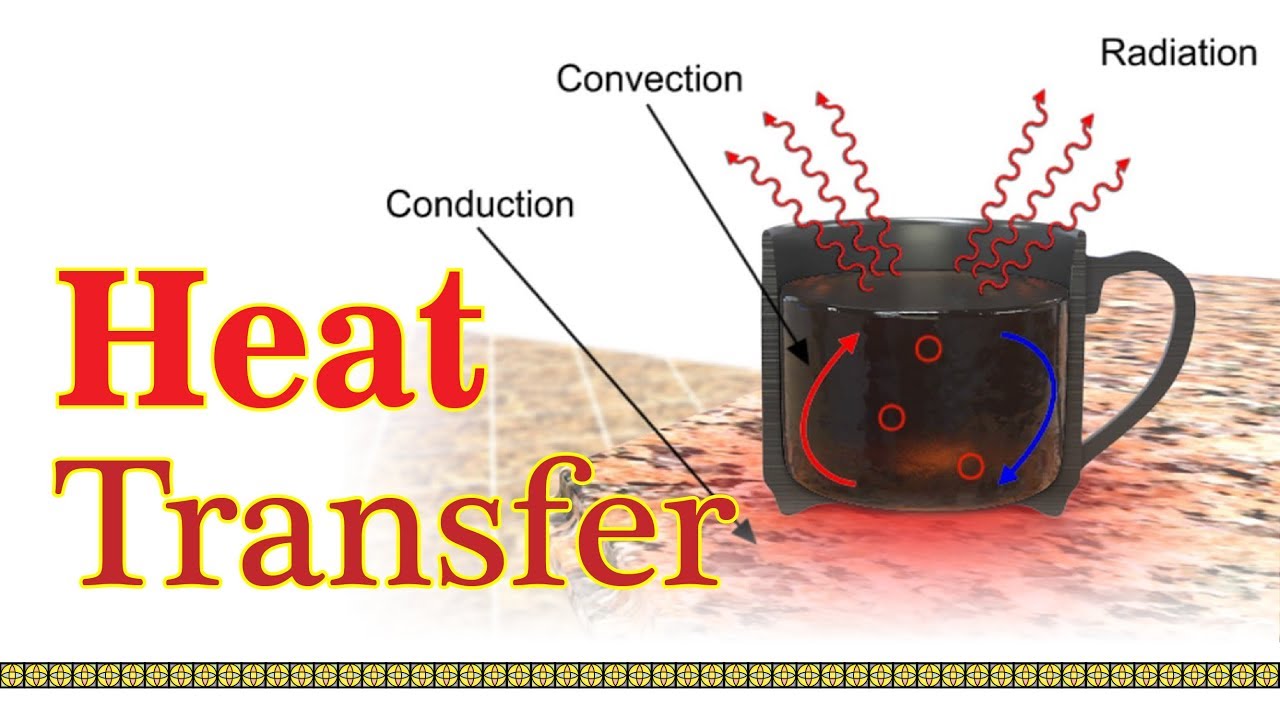
Heat energy can be transmitted from one point to another point in a body by three different methods: conduction, convection and radiation.
Conduction
The process by which heat is transmitted from one point to another particle without actual movement of the particles of the body ( or the medium ) is called conduction.
Conductors and Insulators:
The materials which allow the heat to pass through them easily are called good conductor of heat.
The materials which do not allow the heat to pass through them are called bad conductor of heat.
Conditions for the conduction of heat between bodies:
Convection
The process of transfer of heat in which the heated particles actually move from one place to another place to carry out the heat is convection.
Radiation
The process of transfer of heat from one place to another in absence of medium is called radiation.