↪ Thus obtained metal from above process is not pure and it is further required to carry out purification or refining.
↪ Refining can be done by any of the methods.
↪ The selection of the method depends upon the nature of impurities.
⁕ Electrolytic refining
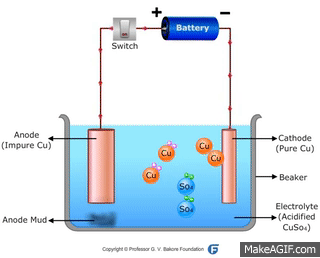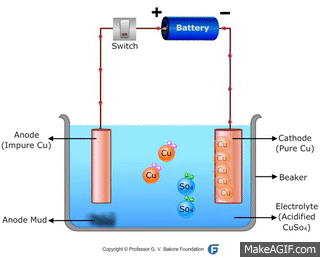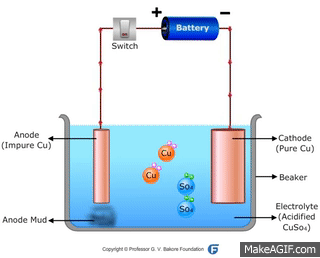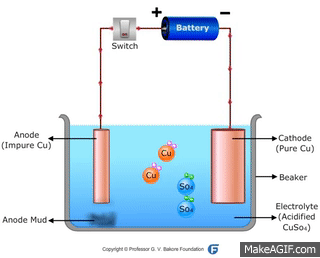
↪ In this method, impure metal is used as anode and pure metal is taken as cathode.
↪ On passing electricity, metal dissolves from anode and equivalent amount of metal gets deposited at cathode.
↪ This method is widely used for metals like Cu, Ag, Au, Zn, Ni etc.
⁕ Poling
↪ This method is useful to remove reduce able oxides from the metal. In this method, crude molten metal is stirred with a green pole of wood.
↪ Hydrocarbon of the pole reduces oxide impurities.

⁕ Liquation
↪ This method is applicable for those metals which have lower melting point than impurities.
↪ Impure metal is placed on slopping furnace and temperature is maintained a bit above the melting point of metal.
↪ The metal melts and flows off of the slope. It is then collected and cooled.
 ⁕ Distillation
⁕ Distillation

↪ In this method, metals having low boiling point (e.g. Zn, Hg and Cd) are refined the metal to be purified is heated at around its boiling point so as to vaporize metal leaving impurities back.
↪ The vapor of the metal is condensed to get pure metal.
Some Terminologies
⇲ Flux
↪ The substance which is added to the ore before heating in order to convert the infusible gangue into light molten mass of slag is called flux.
⇲ Slag
↪ The fusible mass which is obtained by combination of impurities (gangue) and flux is called slag.
↪ Slag is lighter and floats on the surface of molten metal which can be removed separately from metal
Different between flux and Slag
| Flux | Slag |
It is added from out side.
| It is formed by combination of flux and gangue.
|
It is either acidic, basic or neutral.
| It is fusible mass of salt .
|
It is used to remove impurities present in ores.
| Some slag are used fertilizers like Thomas slag, Gilchrist Slag.
|










