Chemical and Physical Equilibrium
↪ A reversible reaction, whether physical or chemical, proceed to a limited extent and both reactants and products are available in the reaction mixture such that the concentrations of reactan...


Equilibrium
↪ The state of a process in which the properties like temperature, pressure, concentration of the system don't show any change with the passage of time is known as Equilibrium.
↪ The equilibrium achieved in physical process is called Physical Equilibrium and that of chemical process is called Chemical Equilibrium.
↪ In a chemical reaction, chemical equilibrium is the state in which both reactants and products are present in constant concentrations which have no further tendency to change with time, so that there is no observable change in the properties of the system.
⁕ Forward Reaction
↪ The reaction having the direction from reactants to products side is called forward reaction.
⁕ Backward reaction
↪ The reaction having the direction from products to reactants side is called backward reaction.
⁕ Reversible Reaction
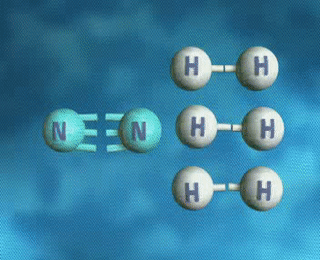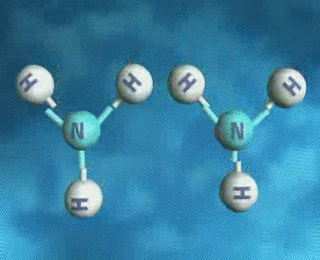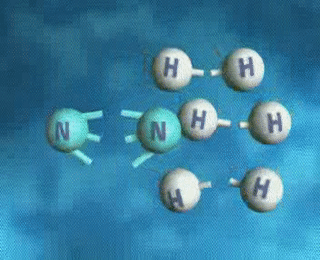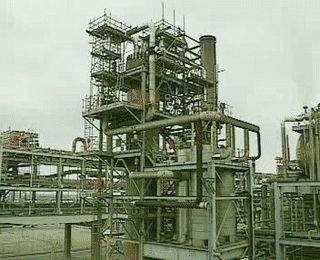
↪ A process is said to be reversible when reactants react to give products and products also react back to give reactants under the same set of conditions.
↪ A reversible process is incomplete and both physical change and chemical reaction may attain it.
↪ It is denoted by double headed arrow ⇌ placed between the reactants and the products.
↪ It is only possible only in closed system or vessel.
↪ It maintains equilibrium state.
For examples:
⁕ Irreversible Reaction
↪ The chemical reaction in which the reactants react to form products but the products don't form reactants is called irreversible reaction.
↪ It is always in a single direction.
↪ It can occur in open vessel or closed vessel.
↪ It does not maintain equilibrium state.
For example: